The Study Hub showcases a collection of groundbreaking clinical trials - many of them world-firsts. They are powered by Spiral’s people and technology to deliver real-world impact and advance global health outcomes.

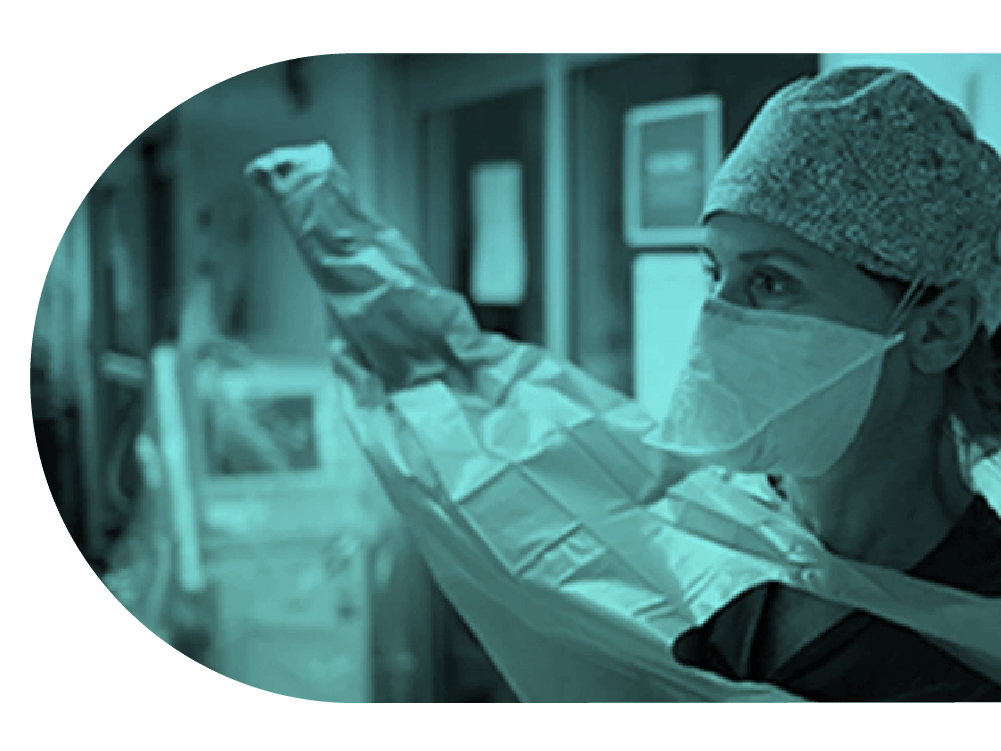
Primary Outcome: Day 90 mortality for pneumonia patients
Trial Outcome: determine the optimal intensive care treatment for patients after arrival in hospital.

Trial Update: An adaptive platform design, running multiple domains concurrently.


Thought leadership

Global delivery, local sovereignty
Data residency isn’t a checkbox; it’s at the core of ethics, regulation, and community trust.

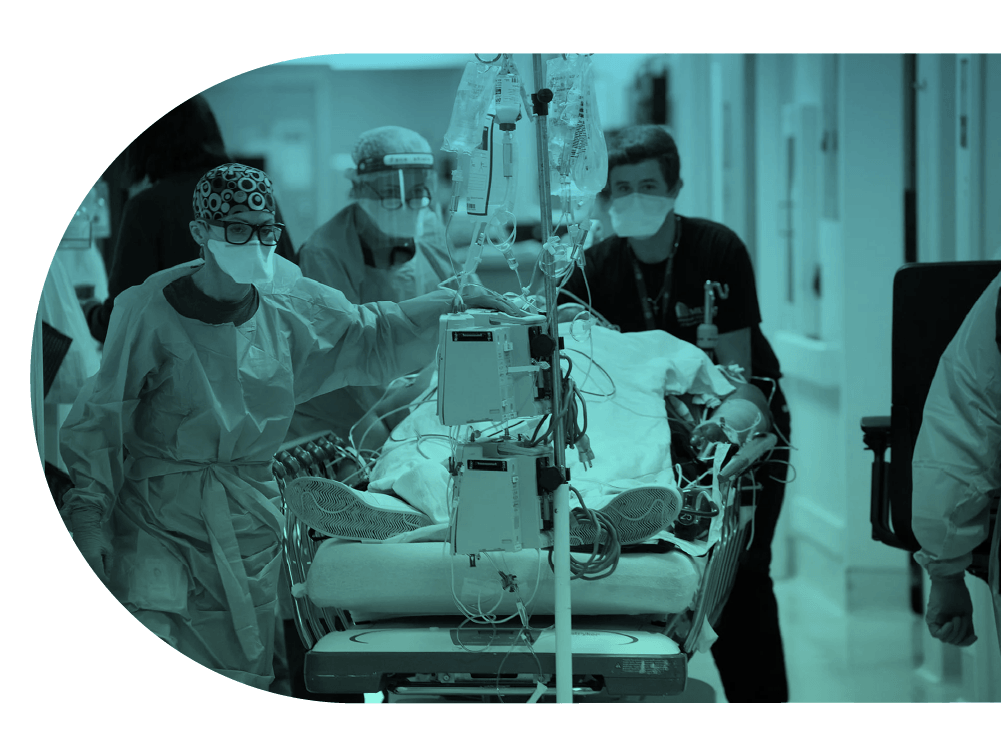
A neonatal trial that spans hospital and intensive care
An Australasian platform trial led out of the University of Melbourne, focused on improving outcomes for babies at risk of preterm birth, or born preterm.

Why checkboxes trip up clinical data
For research nurses, the way a question is built in the CRF can be the difference between a clean lock and a month of queries.
Human-centered design

-

"This trial is about so much more than just numbers – it's about redefining what's possible in healthcare and making a lasting impact on patients' lives."
Diane Mackle, Project Manager, MEGA-ROX Trial
-

“‘In clinical trials, especially academic clinical trials, we don’t often have the luxury of a full system that will do everything for us, normally there would be a separate system for separate things. Spiral provides this full system”.
~Ly-Mee Yu, Lead Statistician, PANORAMIC Trial

Global impact